Efficient 2-step biocatalytic strategies for the synthesis of all nor(pseudo)ephedrine isomers†
Abstract
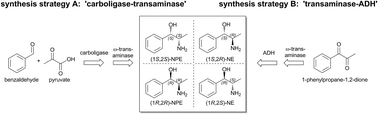
Chiral 1,2-amino alcohols are important building blocks for chemistry and pharmacy. Here, we developed two different biocatalytic 2-step cascades for the synthesis of all four nor(pseudo)ephedrine (N(P)E) stereoisomers. In the first one, the combination of an (R)-selective thiamine diphosphate (ThDP)-dependent carboligase with an (S)- or (R)-selective ω-transaminase resulted in the formation of (1R,2S)-NE or (1R,2R)-NPE in excellent optical purities (ee >99% and de >98%). For the synthesis of (1R,2R)-NPE, space–time yields up to ∼26 g L−1 d−1 have been achieved. Since a highly (S)-selective carboligase is currently not available for this reaction, another strategy was followed to complement the nor(pseudo)ephedrine platform. Here, the combination of an (S)-selective transaminase with an (S)-selective alcohol dehydrogenase yielded (1S,2S)-NPE with an ee >98% and a de >99%. Although lyophilized whole cells are cheap to prepare and were shown to be appropriate for use as biocatalysts, higher optical purities were observed with purified enzymes. These synthetic enzyme cascade reactions render the N(P)E-products accessible from inexpensive, achiral starting materials in only two reaction steps and without the isolation of the reaction intermediates.

 Please wait while we load your content...
Please wait while we load your content...