Porphyrins with a carbosilane dendrimer periphery as synthetic components for supramolecular self-assembly†
Abstract
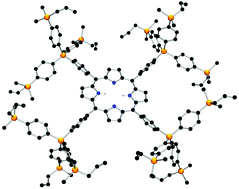
The preparation of the shape-persistent carbosilane-functionalized porphyrins H2TPP(4-SiRR′Me)4, Zn(II)-TPP(4-SiRR′Me)4 (R = R′ = Me, CH2CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2, CH2CH2CH2OH; R = Me, R′ = CH2CH
CH2, CH2CH2CH2OH; R = Me, R′ = CH2CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2, CH2CH2CH2OH; TPP = tetraphenyl porphyrin), H2TPP(4-Si(C6H4-1,4-SiRR′Me)3)4, and Zn(II)-TPP(4-Si(C6H4-1,4-SiRR′Me)3)4 (R = R′ = Me, CH2CH
CH2, CH2CH2CH2OH; TPP = tetraphenyl porphyrin), H2TPP(4-Si(C6H4-1,4-SiRR′Me)3)4, and Zn(II)-TPP(4-Si(C6H4-1,4-SiRR′Me)3)4 (R = R′ = Me, CH2CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2; R = Me, R′ = CH2CH
CH2; R = Me, R′ = CH2CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2) using the Lindsey condensation methodology is described. For a series of five samples their structures in the solid state were determined by single crystal X-ray structure analysis. The appropriate 0th and 1st generation porphyrin-based 1,4-phenylene carbosilanes form 2D and 3D supramolecular network structures, primarily controlled by either π–π interactions (between pyrrole units and neighboring phenylene rings) or directional molecular hydrogen recognition and zinc–oxygen bond formation in the appropriate hydroxyl-functionalized molecules. UV-Vis spectroscopic studies were carried out in order to analyze the effect of the dendritic branches on the optical properties of the porphyrin ring.
CH2) using the Lindsey condensation methodology is described. For a series of five samples their structures in the solid state were determined by single crystal X-ray structure analysis. The appropriate 0th and 1st generation porphyrin-based 1,4-phenylene carbosilanes form 2D and 3D supramolecular network structures, primarily controlled by either π–π interactions (between pyrrole units and neighboring phenylene rings) or directional molecular hydrogen recognition and zinc–oxygen bond formation in the appropriate hydroxyl-functionalized molecules. UV-Vis spectroscopic studies were carried out in order to analyze the effect of the dendritic branches on the optical properties of the porphyrin ring.

 Please wait while we load your content...
Please wait while we load your content...