Synthesis and characterization of β-triketimine cobalt complexes and their behaviour in the polymerization of 1,3-butadiene†
Abstract
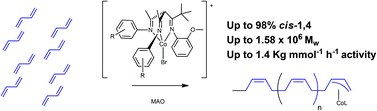
Three β-triketimine ligands, (L1: [CH{CMeN(2,4,6-Me3-C6H2)}2C(CMe3N(2-MeO-C6H4)], L2: [CH{CMeN(2,4-Me2-C6H3)}2C(CMe3N(2-MeO-C6H4)] and L3: [CH{CMeN(2-Me-C6H4)}2C(CMe3N(2-MeO-C6H4)]), were synthesized and then characterized by 1H and 13C{1H} NMR spectroscopy, elemental analysis and electrospray (ES) MS. β-triketimine cobalt(II) complexes were then prepared by the interaction of cobalt(II) bromide with L1–3 in the presence of NaBArF (BArF = [{3,5-(CF3)2-C6H3}4B]− ). Five-coordinate dimeric bromide-bridged species [(LCoμ-Br)2][BArF]2 were obtained. The geometry of the complexes was found by single-crystal X-ray diffraction to be slightly distorted square-pyramidal. The polymerization of 1,3-butadiene catalysed by these complexes upon activation with methylaluminoxane (MAO) in chlorobenzene yielded high cis-1,4-polybutadiene (>97% cis). The effect of steric and electronic properties of the ligands on the performance of the catalytic system was investigated: it was found that ligands with fewer methyl substituents gave more active catalytic systems. It was also found that increasing MAO: Co ratio resulted in higher activity. Stereoselectivity of all catalysts slightly decreased at higher temperature, whereas activities were maximised at 20 °C, where very high values of activity were recorded.

 Please wait while we load your content...
Please wait while we load your content...