Redox-exchange induced heterogeneous RuO2-conductive polymer nanowires†
Abstract
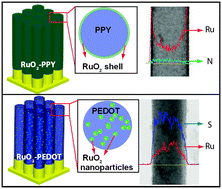
A redox exchange mechanism between potassium perruthenate (KRuO4) and the functional groups of selected polymers is used here to induce RuO2 into and onto conductive polymer nanowires by simply soaking the polymer nanowire arrays in KRuO4 solution. Conductive polymer nanowire arrays of polypyrrole (PPY) and poly(3,4-ethylenedioxythiophene) (PEDOT) were studied in this work. SEM and TEM results show that the RuO2 material was distributed differently in the PPY and PEDOT nanowire matrices. Energy-dispersive X-ray spectroscopy and X-ray photoelectron spectroscopy were used to confirm the dispersion and formation of RuO2 materials in these polymer nanowires. Cyclic voltammetry and galvanostatic charge–discharge experiments were used to characterize their electrochemical performance. RuO2–polymer samples prepared with a 6 min soaking time in 10 mM KRuO4 solution show a high specific capacitance of 371 F g−1 and 500 F g−1 for PEDOT-based and PPY-based composite nanowires, respectively. This is attributed to the high exposure area of the conductive RuO2 and the good conductivity of the polymer matrix. This work demonstrates a simple method to synthesize heterogeneous polymer based-materials through the redox reaction between conductive polymers and high oxidation state transition metal oxide ions. Different heterogeneous nanocomposites were obtained depending on the polymer properties, and high energy storage performance of the metal oxides can be achieved within these heterogeneous nanostructures.

 Please wait while we load your content...
Please wait while we load your content...