Effect of ion structure on nanoscale friction in protic ionic liquids†
Abstract
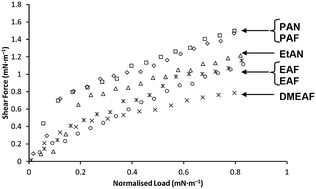
The effect of ionic liquid (IL) molecular structure on nanoscale friction has been investigated using colloidal probe Friction Force Microscopy (FFM). The ILs studied were ethylammonium formate (EAF), ethylammonium nitrate (EAN), propylammonium formate (PAF), propylammonium nitrate (PAN), dimethylethylammonium formate (DMEAF), and ethanolammonium nitrate (EtAN). ILs were confined between a silica colloid probe and a mica surface, and the friction force was measured as a function of normal load for sliding velocities between 10 and 40 μm s−1. At low normal forces, multiple IL layers are found between the probe and the surface, but at higher force, in the boundary layer regime, a single ion layer separates the probe and the surface. In the boundary layer regime energy is dissipated by two main pathways. Firstly, the ionic liquid near the surface, with the exception of the boundary layer, is expelled from the advancing contact made by the probe on the surface. This disruption in the interactions between the boundary layer and the near surface multilayers, leads to energy dissipation and depends on the strength of the attraction between the boundary and near surface layers. The second pathway is via rotations and twists of ions in the boundary layer, primarily associated with the cation terminal methyl group. The friction coefficient did not vary over the limited range of sliding speeds investigated.

 Please wait while we load your content...
Please wait while we load your content...