Structures and properties of coordination polymers involving asymmetric biphenyl-3,2′,5′-tricarboxylate†
Abstract
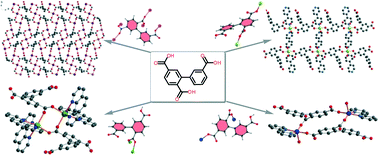
An asymmetric polycarboxylate ligand, biphenyl-3,2′,5′-tricarboxylic acid (H3bptc) with a rotatable coordination vertex, was employed to react with metal ions under hydrothermal conditions. Four new coordination polymers {[Mn3(bptc)2(4,4′-bpy)3(H2O)2]}n (1), {[Ni(Hbptc)(4,4′-bpy)1.5(H2O)2]·H2O}n (2), {[Cd2(Hbptc)2(phen)4]·3H2O}n (3) and {[Cu2(Hbptc)2(2,2′-bpy)2(H2O)2]·2H2O}n (4) were successfully synthesized and characterized. Single crystal X-ray analysis revealed that compound 1 has a 3D coordination network with linear Mn(II)3-based subunits, while 2 is a 1D ladder chain-based structure further interconnected by hydrogen bonds to a 3D supramolecular network. Compounds 3 and 4 are each composed of a supramolecular network, which is supported by hydrogen bonds and π⋯π interactions and consists of dimers, [Cd2(Hbptc)2(phen)4] and [Cu2(Hbptc)2(2,2′-bpy)2], respectively. The conformation stability and coordination modes of the H3bptc ligand have been discussed. The magnetic and fluorescent properties of the complexes have been investigated.

 Please wait while we load your content...
Please wait while we load your content...