A gold-catalysed fully intermolecular oxidation and sulfur-ylide formation sequence on ynamides†
Abstract
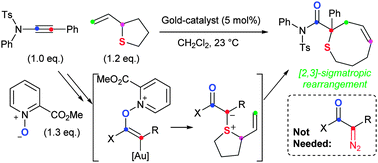
An efficient C–O, C–S and C–C bond-forming sequence leads to functionalised compounds bearing sulfur-substituted quaternary carbons. Ynamides are employed as diazo-equivalents to access the [2,3]-sigmatropic rearrangements of allyl sulfonium ylides by a three-component chemoselective oxidation and intermolecular ylide formation.

 Please wait while we load your content...
Please wait while we load your content...