Two-directional synthesis and biological evaluation of alkaloid 5-epi-cis-275B′†
Abstract
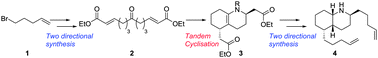
The first total synthesis of myrmicine ant alkaloid 5-epi-cis-275B′ (4) is presented. A tandem cyclisation established the entire core of the structure in a single transformation as well as the required 2,5-anti stereochemistry. Two-directional synthesis was used to furnish the cyclisation precursor 2, as in each of the subsequent steps towards the natural product. The first electrophysiology studies for 4 (against nicotinic acetylcholine receptors) were also conducted, finding modest inhibition of current.


 Please wait while we load your content...
Please wait while we load your content...