Fabrication of novel chemosensors composed of rhodamine derivative for the detection of ferric ion and mechanism studies on the interaction between sensor and ferric ion†
Abstract
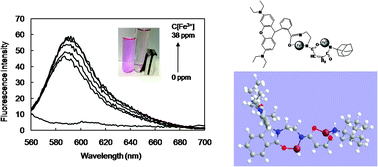
Although many rhodamine based fluorescence sensors were reported to detect metal ions with high sensitivity and selectivity, there are very few reports available to study the mechanisms of detection and the interaction between probe and metal ions. This paper aims to detect ferric ions by novel fluorescence chemosensors and study the mechanisms in detail. A novel probe AD-MAH-RhB was designed and synthesized from rhodamine B (RhB), adamantyl (AD), ethylene diamine and maleic anhydride (MAH). AD-MAH-RhB could detect Fe3+ in aqueous solutions. The mechanism was explored by the HSAB principle, FTIR and mass spectra. The results suggested that Fe3+ bound with amine and oxygen atoms in AD-MAH-RhB to form a complex composed of a 2 : 1 stoichiometry of Fe3+ and the probe. Moreover, computational simulations were employed to further investigate the detection mechanism. The calculated results showed that Fe3+ could conjugate with AD-MAH-RhB probe to form a stable complex, which was induced by synergetic effects of the suitable space and distance of van der Waals forces. However, Hg2+ was found to disturb this detection and form a complex with 1 : 2 stoichiometry of Hg2+ and AD-MAH-RhB. Then, another probe, β-cyclodextrin modified polymaleic anhydride (PMAH-CD) including AD-MAH-RhB (PMAH-CD/AD-MAH-RhB) was fabricated by inclusion interaction between CD and AD. PMAH-CD@AD-MAH-RhB showed high selectivity and sensitivity to Fe3+ in the aqueous solution by eliminating the interruption of Hg2+ possibly due to the high hydrogen interaction among the probes to inhibit the stable form complex with Hg2+.

 Please wait while we load your content...
Please wait while we load your content...