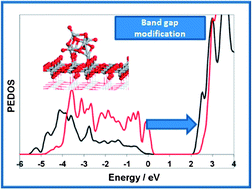
Titanium dioxide is an important photocatalytic material with much activity in modifying it to achieve visible light absorption. Recently, studies targeting the modification of TiO2 with metal oxide nano clusters have investigated heterostructures which show potential as new TiO2-based photocatalytic materials with visible light absorption and improved photocatalytic activity. Obtaining detailed insights into the influence of surface modification on the photocatalytic properties of TiO2 will further the possibilities for using this approach to develop new photocatalysts. In this paper, we present the results of density functional theory (DFT) simulations of the TiO2 rutile (110) surface modified with TiO2 nanoclusters ((TiO2)n: Ti5O10, Ti6O12, Ti8O16, Ti16O32, Ti30O60), with diameters up to 1.5 nm, which is a cluster size achievable in experiments. The clusters adsorb strongly at the surface giving adsorption energies from −2.7 eV to −6.7 eV. The resulting structures show a large number of new Ti–O bonds created between the cluster and the surface, so that the new bonds enhance the stability. The electronic density of states (EDOS) shows that the valence band edge is shifted upwards, due to new nanocluster derived states in this region, while the conduction band is unchanged and composed of the Ti 3d states from the surface. This reduces the band gap over bare TiO2, potentially shifting light absorption into the visible region. The valence and conduction band composition of these heterostructures will favour spatial separation of electrons and holes after light excitation, thus giving improved photocatalytic properties in these novel structures.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...