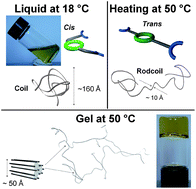
We present the aggregation mechanism in a novel class of heat-set metallosupramolecular Ni2BTC gels formed by complexation of a bis-terpyridine-cyclam (BTC), a tritopic ligand, with nickel ions (II). Viscosimetry, rheology, small-angle X-ray scattering, dynamic light scattering and impedance spectroscopy measurements are used to investigate the aggregative properties. Gels are obtained by heating the homogeneous solutions (Ni2BTC in dimethylformamide) from room temperature to 50 °C for 24 hours, which is a remarkable phenomenology in the class of organic molecular gels. The hierarchical aggregation mechanism relies on a temperature-triggered cis-to-trans molecular conversion of the tritopic ligand at the central cyclam unit. At the nanoscale, the expansion of the coil-like species in the liquid phase to rodcoils allows, beyond a critical concentration, their percolation into transient gel networks. The use of such tritopic ligands gives access to homo- or hetero-metallogels that adapt their properties to the environmental conditions (temperature, pH, mechanical stress and applied electrical potential). Various molecular adjustment parameters (metal ions, metal sequence, metal–ligand stoichiometry, cyclam substituents) can be used to tune the aggregative properties of the system.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...