Gelation of the genome by topoisomerase II targeting anticancer agents†
Abstract
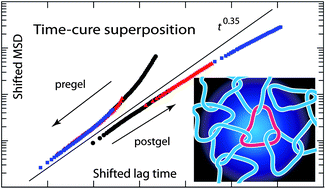
Topoisomerase II (TOP2) regulates the topology of DNA by catalysis of a double strand passage reaction. Inhibition of this reaction prevents cell replication, and, thus, is a pathway targeted by anticancer drugs. Some details regarding the cell-killing mechanism are unknown and assays to screen for anticancer drugs are not well established. Here, we study the gelation of linear and circular DNA using microrheology assays. Gelation of the DNA–enzyme mixture was examined by tracking of multiple colloidal probe particles. The mean square displacements of the probe particles were analyzed by the time-cure superposition procedure as well as the classical derivation of the dynamic moduli. First, the passage reaction was inhibited by AMP-PNP, a non-hydrolyzable analog of ATP. The results showed gelation due to the formation of a self-catenated network of circular DNA molecules. Next, when TOP2 was inhibited by the anti-cancer drug ICRF-193, we observed a similar change in rheology. Based on these findings, we propose a cell-killing mechanism by gelation of the genome through TOP2-mediated interlocking of looped DNA segments of the replicated, intertwined chromosomes.

 Please wait while we load your content...
Please wait while we load your content...