Photoactively electroactive polyamide with azo group in the main chain via oxidative coupling polymerization
Abstract
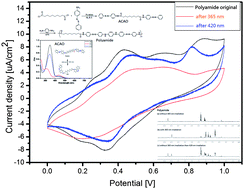
In this work, a simultaneously photoactive and electroactive polyamide exhibiting interesting spectroscopic and electrochemical properties was prepared. Firstly, a 4-aminoazobenzene-capped amide oligomer (ACAO) was synthesized and characterized by 1H NMR, 13C NMR and mass spectroscopy. Subsequently, the photoactively electroactive polyamide was synthesized through reacting ACAO with p-phenylenediamine by oxidative coupling polymerization, followed by characterization with GPC and 1H NMR. After alternate irradiation with UV and visible light, the azobenzene exhibited obvious photoelectrochemical switching properties. The reversible photoisomerization (i.e., cis ↔ trans) behaviour of the backbone azobenzene moieties in the as-prepared photoactively electroactive polyamide was analyzed by in situ monitoring through systematic studies of UV-visible and 1H NMR spectroscopies. On the other hand, the electrochemical activity (i.e., redox capability) of the as-prepared polyamide was explored by cyclic voltammetry (CV) measurements.

 Please wait while we load your content...
Please wait while we load your content...