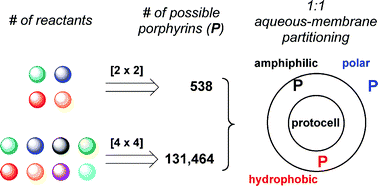
The role of combinatorial processes in the origin of life remains relatively unexplored. In a chemical model for the possible prebiogenesis of tetrapyrrole macrocycles reported previously, a tandem combinatorial reaction of two diones (substituents = methyl, acetic acid) and two aminoketones (substituents = ethyl, propanoic acid) afforded up to 538 porphyrins (upon oxidation of the corresponding porphyrinogens). The reaction was performed at a 1 : 1 ratio of hydrophobic and hydrophilic substituents in each pool of reactants, and the resulting porphyrins partitioned in ∼1 : 1 ratio between aqueous solution and phosphatidylcholine vesicle membranes. Here, a change in the ratio of hydrophobic and hydrophilic substituents of the [2 × 2] reaction gave corresponding changes in the polarity profile of the resulting porphyrins (3.5–9.0% yield). Reaction of four diones and four aminoketones (bearing hydrophilic or hydrophobic substituents) in the presence of lipid vesicles followed by photooxidation afforded porphyrins in 8.7% yield. The resulting porphyrins partitioned in ∼1 : 1 ratio between phosphatidylcholine vesicles and aqueous solution, as observed previously for the [2 × 2] reaction. Both the aqueous fraction and the vesicles fraction were photochemically active as evidenced by the fluorescence quantum yield (Φf ∼ 0.1). Software (PorphyrinViLiGe) for virtual library generation indicates that the [4 × 4] reaction affords up to 131 464 porphyrins. The relative insensitivity of physicochemical properties (partitioning, photoactivity) toward combinatorial expansion may be a valuable yet unappreciated attribute for prebiotic functionality.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...