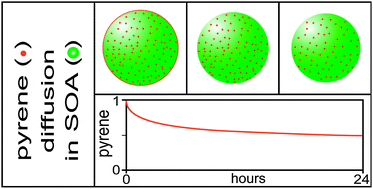
Formation, properties, transformations, and temporal evolution of secondary organic aerosol (SOA) particles depend strongly on SOA phase. Recent experimental evidence from both our group and several others indicates that, in contrast to common models' assumptions, SOA constituents do not form a low-viscosity, well-mixed solution, yielding instead a semisolid phase with high, but undetermined, viscosity. We find that when SOA particles are made in the presence of vapors of semi-volatile hydrophobic compounds, such molecules become trapped in the particles' interiors and their subsequent evaporation rates and thus their rates of diffusion through the SOA can be directly obtained. Using pyrene as the tracer molecule and SOA derived from α-pinene ozonolysis, we find that it takes ∼24 hours for half the pyrene to evaporate. Based on the observed pyrene evaporation kinetics we estimate a diffusivity of 2.5 × 10−21 m2 s−1 for pyrene in SOA. Similar measurements on SOA doped with fluoranthene and phenanthrene yield diffusivities comparable to that of pyrene. Assuming a Stokes–Einstein relation, an approximate viscosity of 108 Pa s can be calculated for this SOA. Such a high viscosity is characteristic of tars and is consistent with published measurements of SOA particle bounce, evaporation kinetics, and the stability of two reverse-layered morphologies. We show that a viscosity of 108 Pa s implies coalescence times of minutes, consistent with the findings that SOA particles formed by coagulation are spherical on the relevant experimental timescales. Measurements on aged SOA particles doped with pyrene yield an estimated diffusivity ∼3 times smaller, indicating that hardening occurs with time, which is consistent with the increase in SOA oligomer content, decrease in water uptake, and decrease in evaporation rates previously observed with aging.

 Please wait while we load your content...
Please wait while we load your content...