Temperature-induced phase separation and hydration in poly(N-vinylcaprolactam) aqueous solutions: a study by NMR and IR spectroscopy, SAXS, and quantum-chemical calculations
Abstract
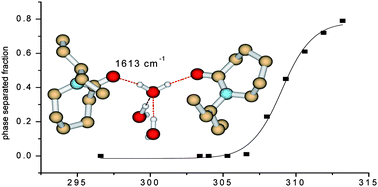
Temperature-induced phase separation and hydration in poly(N-vinylcaprolactam) (PVCL)–D2O solutions were investigated by a combination of

 Please wait while we load your content...
Please wait while we load your content...