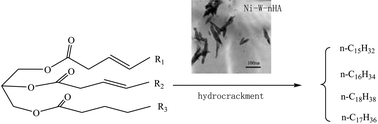
The synthesis, characterization and catalytic capability of the NiW–nano-hydroxyapatite (NiW–nHA) composite were investigated in this paper. The NiW–nHA catalyst was prepared by a co-precipitation method. Then Fourier transform infrared (FT-IR) spectroscopy, transmission electron microscopy (TEM), X-ray diffraction (XRD) and energy dispersive spectroscopy (EDX) were used to analyze this material. In addition, the catalytic capacity of the NiW–nHA composite was also examined by FT-IR and gas chromatography (GC). The results of FT-IR analysis indicated that Ni, W and nHA combined closely. TEM observation revealed that this catalyst was needle shaped and the crystal retained a nanometer size. XRD data also suggested that a new phase of CaWO4 appeared and the lattice parameters of nHA changed in this system. nHA was the carrier of metals. The rates of Ni/W-loading were 73.24% and 65.99% according to the EDX data, respectively. Furthermore, the conversion of 91.88% Jatropha oil was achieved at 360 °C and 3 MPa h−1 over NiW–nHA catalyst. The straight chain alkanes ranging from C15 to C18 were the main components in the production. The yield of C15–C18 alkanes was up to 83.56 wt%. The reaction pathway involved hydrocracking of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds of these triglycerides from Jatropha oil. This paper developed a novel non-sulfided catalyst to obtain a “green biofuel” from vegetable oil.
C bonds of these triglycerides from Jatropha oil. This paper developed a novel non-sulfided catalyst to obtain a “green biofuel” from vegetable oil.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds of these
C bonds of these 

 Please wait while we load your content...
Please wait while we load your content...