Electrophilic natural products and their biological targets
Abstract
Covering: 1900 to 2011
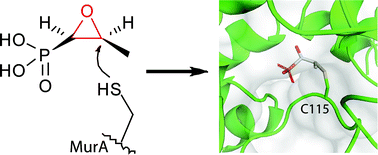
The study of biologically active natural products has resulted in seminal contributions to our understanding of living systems. In the case of electrophilic natural products, the covalent nature of their interaction has largely facilitated the identification of their biological binding partners. In this review, we provide a comprehensive compilation of electrophilic natural products from all major chemical classes together with their biological targets. Covering Michael acceptor systems, ring-strained compounds and other electrophiles, such as esters or carbamates, we highlight representative and instructive examples for over 20 electrophilic moieties. The fruitful cooperation of natural product chemistry, medicinal chemistry and chemical biology has produced a collection of well-studied examples for how electrophilic natural products exert their biological functions that range from antibiotic to antitumor effects. Special emphasis is put on the elucidation of their respective biological targets via activity-based protein profiling, which together with the recent advancements in mass spectrometry has been crucial to the success of the field. The wealth of naturally occurring electrophilic moieties and their chemical complexity enables binding of a large variety of biological targets, such as enzymes of all classes, nonenzymatic proteins, DNA and other cellular compounds. With approximately 30 000 genes in the human genome but only 266 confirmed protein drug targets, the study of biologically active, electrophilic natural products has the potential to provide insights into fundamental biological processes and to greatly aid the discovery of new drug targets.

 Please wait while we load your content...
Please wait while we load your content...