Integrated separation of blood plasma from whole blood for microfluidic paper-based analytical devices
Abstract
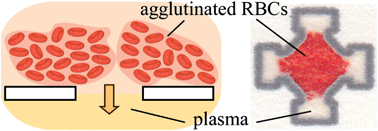
Many diagnostic tests in a conventional clinical laboratory are performed on blood plasma because changes in its composition often reflect the current status of pathological processes throughout the body. Recently, a significant research effort has been invested into the development of microfluidic paper-based analytical devices (μPADs) implementing these conventional laboratory tests for point-of-care diagnostics in resource-limited settings. This paper describes the use of red blood cell (RBC) agglutination for separating plasma from finger-prick volumes of whole blood directly in paper, and demonstrates the utility of this approach by integrating plasma separation and a colorimetric assay in a single μPAD. The μPAD was fabricated by printing its pattern onto

 Please wait while we load your content...
Please wait while we load your content...