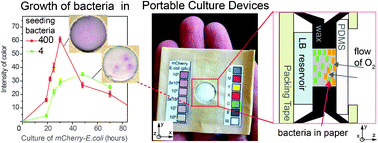
In this paper, we demonstrate that a functional, portable device for the growth of bacteria or amplification of bacteriophage can be created using simple materials. These devices are comprised of packing tape, sheets of paper patterned by hydrophobic printer ink, and a polydimethyl siloxane (PDMS) membrane, which is selectively permeable to oxygen but non-permeable to water. These devices supply bacteria with oxygen and prevent the evaporation of media for a period over 48 h. The division time of E. coli and the amplification of the phage M13 in this device are similar to the rates measured on agar plates and in shaking cultures. The growth of bacteria with a fluorescent mCherry reporter can be quantified using a flatbed scanner or a cell phone camera. Permeating devices with commercial viability dye (PrestoBlue) can be used to detect low copy number of E. coli (1–10 CFU in 100 μL) and visualize microorganisms in environmental samples. The platform, equipped with bacteria that carry inducible mCherry reporter could also be used to quantify the concentration of the inducer (here, arabinose). Identical culture platforms can, potentially, be used to quantify the induction of gene expression by an engineered phage or by synthetic transcriptional regulators that respond to clinically relevant molecules. The majority of measurement and fabrication procedures presented in this report have been replicated by low-skilled personnel (high-school students) in a low-resource environment (high-school classroom). The fabrication and performance of the device have also been tested in a low-resource laboratory setting by researchers in Nairobi, Kenya. Accordingly, this platform can be used as both an educational tool and as a diagnostic tool in low-resource environments worldwide.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...