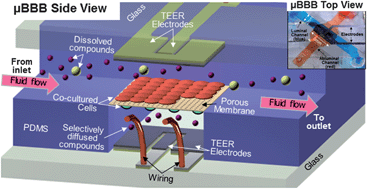
The blood-brain barrier (BBB), a unique selective barrier for the central nervous system (CNS), hinders the passage of most compounds to the CNS, complicating drug development. Innovative in vitro models of the BBB can provide useful insights into its role in CNS disease progression and drug delivery. Static transwell models lack fluidic shear stress, while the conventional dynamic in vitro BBB lacks a thin dual cell layer interface. To address both limitations, we developed a microfluidic blood-brain barrier (μBBB) which closely mimics the in vivo BBB with a dynamic environment and a comparatively thin culture membrane (10 μm). To test validity of the fabricated BBB model, μBBBs were cultured with b.End3 endothelial cells, both with and without co-cultured C8-D1A astrocytes, and their key properties were tested with optical imaging, trans-endothelial electrical resistance (TEER), and permeability assays. The resultant imaging of ZO-1 revealed clearly expressed tight junctions in b.End3 cells, Live/Dead assays indicated high cell viability, and astrocytic morphology of C8-D1A cells were confirmed by ESEM and GFAP immunostains. By day 3 of endothelial culture, TEER levels typically exceeded 250 Ω cm2 in μBBB co-cultures, and 25 Ω cm2 for transwell co-cultures. Instantaneous transient drop in TEER in response to histamine exposure was observed in real-time, followed by recovery, implying stability of the fabricated μBBB model. Resultant permeability coefficients were comparable to previous BBB models, and were significantly increased at higher pH (>10). These results demonstrate that the developed μBBB system is a valid model for some studies of BBB function and drug delivery.

 Please wait while we load your content...
Please wait while we load your content...