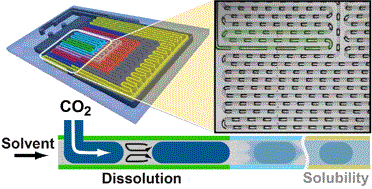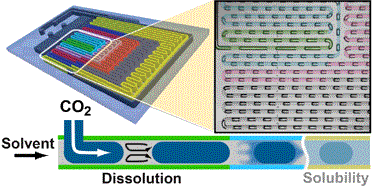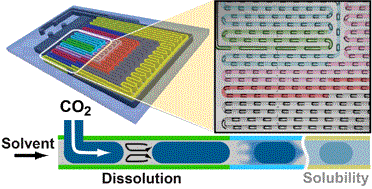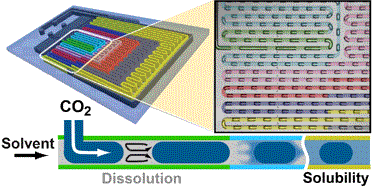
We present an automated microfluidic (MF) approach for the systematic and rapid investigation of carbon dioxide (CO2) mass transfer and solubility in physical solvents. Uniformly sized bubbles of CO2 with lengths exceeding the width of the microchannel (plugs) were isothermally generated in a co-flowing physical solvent within a gas-impermeable, silicon-based MF platform that is compatible with a wide range of solvents, temperatures and pressures. We dynamically determined the volume reduction of the plugs from images that were accommodated within a single field of view, six different downstream locations of the microchannel at any given flow condition. Evaluating plug sizes in real time allowed our automated strategy to suitably select inlet pressures and solvent flow rates such that otherwise dynamically self-selecting parameters (e.g., the plug size, the solvent segment size, and the plug velocity) could be either kept constant or systematically altered. Specifically, if a constant slug length was imposed, the volumetric dissolution rate of CO2 could be deduced from the measured rate of plug shrinkage. The solubility of CO2 in the physical solvent was obtained from a comparison between the terminal and the initial plug sizes. Solubility data were acquired every 5 min and were within 2–5% accuracy as compared to literature data. A parameter space consisting of the plug length, solvent slug length and plug velocity at the microchannel inlet was established for different CO2–solvent pairs with high and low gas solubilities. In a case study, we selected the gas–liquid pair CO2–dimethyl carbonate (DMC) and volumetric mass transfer coefficients 4–30 s−1 (translating into mass transfer times between 0.25 s and 0.03 s), and Henry's constants, within the range of 6–12 MPa.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...