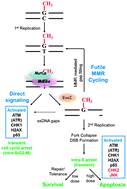
The O6-methylguanine (O6MeG) DNA lesion is well known for its mutagenic, carcinogenic, and cytotoxic properties, and understanding how a cell processes such damage is of critical importance for improving current cancer therapy. Here we use human cells differing only in their O6MeG DNA methyltransferase (MGMT) or mismatch repair (MMR) status to explore the O6MeG/MMR-dependent molecular and cellular responses to treatment with the methylating agent N-methyl-N′-nitro-N-nitrosoguanidine (MNNG). We find that O6MeG triggers MMR-dependent cell cycle perturbations in both the first and second cell cycle post treatment. At lower levels of damage, we show that a transient arrest in the second S-phase precedes survival and progression into subsequent cell cycles. However, at higher levels of damage, arrest in the second S-phase coincides with a cessation of DNA replication followed by initiation of apoptotic cell death. Further, we show that entry into apoptotic cell death is specifically from S-phase of the second cell cycle. Finally, we demonstrate the key role of an O6MeG/MMR-dependent multi-pathway, multi-time-scale signaling network activation, led by early ATM, H2AX, CHK1, and p53 phosphorylation and followed by greatly amplified late phosphorylation of the early pathway nodes along with activation of the CHK2 kinase and the stress-activated JNK kinase.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...