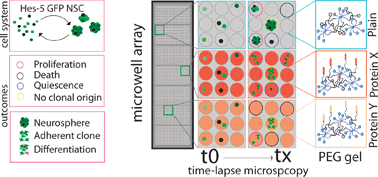
To better understand the extrinsic signals that control neural stem cell (NSC) fate, here we applied a microwell array platform which allows high-throughput clonal analyses of NSCs, cultured either as neurospheres or as adherent clones, exposed to poly(ethylene glycol) (PEG) hydrogel substrates functionalized with selected signaling molecules. We analyzed by time-lapse microscopy and retrospective immunostaining the role of integrin and Notch ligands, two key NSC niche components, in altering the behavior of several hundred single stem cells isolated from a previously described Hes5::GFP reporter mouse. NSC self-renewal was increased by 1.5-fold upon exposure to covalently tethered Laminin-1 and fibronectin fragment 9–10 (FN9–10), where 60–65% of single cells proliferated extensively and remained Nestin positive. Tethering of the Notch ligand Jagged-1 induced activation of Notch signaling. While Jagged-1 alone increased cell survival and proliferation, no further increase in the clonogenic potential of Hes5::GFP cells was observed upon co-stimulation with Laminin-1 and Jagged-1. We believe that the bioengineering of such in vitro niche analogues is a powerful approach to elucidate single stem cell fate regulation in a well-controlled fashion.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...