Template-free synthesis of an electroactive Au-Calix-PPY nanocomposite for electrochemical sensor applications†
Abstract
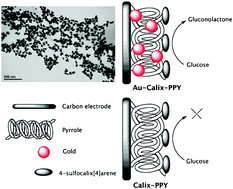
A simple green approach to the template-free synthesis of a nanocomposite material accompanied with a simultaneous redox reaction is described herein. Gold (21.35%) and polypyrrole doped with 4-sulfocalix[4]arene consist in the as-obtained nanobeads with a uniform size in the range of 30 to 50 nm. Universal solvent (water) was selected for the synthesis, allowing the formation of the nanocomposite when the fast addition of the above three components takes place one after the other. The effects of reaction time, temperature, and the concentrations of the dopant, 4-sulfocalix[4]arene and gold on the synthesis of the nanocomposite were also investigated. The current work focuses on the highly electroactive nanocomposite which can be synthesized in the absence of an external additive/template; and from the green chemistry approach, where HAuCl4 serves as an oxidizing agent as well as a stabilizer for the polymer, which makes the synthesis more constructive to avoid the use of hazardous organic solvents. For comparison, we also synthesized polypyrrole (PPY) doped with 4-sulfocalix[4]arene (Calix-PPY), but the synthesis was carried out in the presence of the oxidizing agent (ammonium persulfate), that endorses polymerization of the pyrrole. The Au-Calix-PPY modified electrodes have long-time stability for sensing formaldehyde and glucose. The materials synthesized herein were characterized by UV-visible spectroscopy, FT-IR spectroscopy, Raman spectroscopy, scanning electron microscopy (SEM), transmission electron microscopy (TEM), dynamic light scattering (DLS), energy-dispersive X-ray spectroscopy (EDX) and X-ray photoelectron spectroscopy (XPS). It was revealed that the resulting Au-Calix-PPY nanocomposite exhibits significant electrochemical activity and is successfully demonstrated in sensing formaldehyde and glucose, respectively.

 Please wait while we load your content...
Please wait while we load your content...