Experimental and theoretical studies of the acid-catalyzed conversion of furfuryl alcohol to levulinic acid in aqueous solution†
Abstract
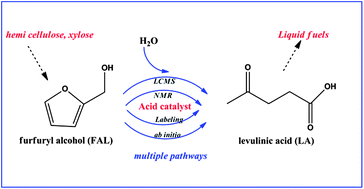
The conversion of furfuryl alcohol (FAL) to levulinic acid over AmberlystTM 15 in aqueous media was investigated using a combination of liquid chromatography-mass spectrometry (LC-MS) measurements, isotopic labeling studies, nuclear magnetic resonance (NMR) spectroscopy, and ab initio quantum chemical calculations using the G4MP2 method. The results of these combined studies showed that one of the major reaction pathways takes place via a geminal diol species (4,5,5-trihydroxypentan-2-one, denoted as intermediate A), formed by the addition of two water molecules to FAL, where two of the oxygen atoms from FAL are retained. This geminal diol species can also be produced from another intermediate found to be a dimer-like species, denoted as intermediate B. This dimer-like species is formed at the early stages of reaction, and it can also be converted to intermediate A, indicating that intermediate B is the product of the reaction of FAL with another early intermediate. Quantum chemical calculations suggested this to be a protonated acyclic species. Reaction of this early intermediate with water produces intermediate A, while reaction with FAL produces intermediate B.

 Please wait while we load your content...
Please wait while we load your content...