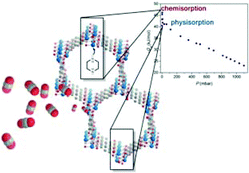
The metal–organic framework Ni2(dobdc) (CPO-27-Ni, where dobdc = 1,4-dioxido-2,5-benzenedicarboxylate) has been post-synthetically modified with piperazine (pip) – a known ‘accelerator’ to improve the kinetics of CO2 uptake in alkanolamine solvents for chemical absorption – and the impact of the modification on the CO2 uptake and selectivity over N2 has been probed. While the modified framework, Ni2(dobdc)(pip)0.5 (pip-CPO-27-Ni), exhibits a lower uptake of CO2 compared with the non-grafted material, the selectivity for CO2 over N2 at 25 °C and at pressures pertinent to post-combustion flue gas capture (0.1–0.15 bar) is enhanced. Mechanistically, the interaction between the CO2 molecules and the free amine sites in pip-CPO-27-Ni occurs via physisorption and chemisorption interactions, in which CO2 binds to the framework with an isosteric heat of adsorption (−Qst) of 40.5 kJ mol−1 at very low coverage (P = 0.033 mbar), followed by binding at a higher heat of adsorption (−Qst = 46.2 kJ mol−1 at P = 3.55 mbar). Pure water adsorption isotherms revealed a two-step mechanism for uptake in CPO-27-Ni, consistent with adsorption into the first and second hydration spheres of Ni2+ followed by subsequent uptake via physisorption into the pores. Additional steric hindrance in pip-CPO-27-Ni results in a single step only. The working capacity over multiple cycles was also investigated using a temperature swing adsorption process which revealed reversible CO2 adsorption and desorption of 10 wt% over 10 cycles.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...