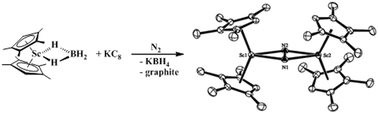
The synthetically accessible borohydride complexes (C5Me4H)2Ln(THF)(BH4) and (C5Me5)2Ln(THF)(BH4) (Ln = Sc, Y) were examined as precursors alternative to the heavily-used tetraphenylborate analogs, [(C5Me4H)2Ln][BPh4] and [(C5Me5)2Ln][BPh4], employed in LnA2A′/M reduction reactions (A = anion; M = alkali metal) that generate “LnA2” reactivity and form reduced dinitrogen complexes [(C5R5)2(THF)xLn]2(μ-η2:η2-N2) (x = 0, 1). The crystal structures of the yttrium borohydrides, (C5Me4H)2Y(THF)(μ-H)3BH, 1, and (C5Me5)2Y(THF)(μ-H)2BH2, 2, were determined for comparison with those of the yttrium tetraphenylborates, [(C5Me4H)2Y][(μ-Ph)2BPh2], 3, and [(C5Me5)2Y][(μ-Ph)2BPh2], 4. The complex (C5Me4H)2Sc(μ-H)2BH2, 5, was synthesized and structurally characterized for comparison with (C5Me5)2Sc(μ-H)2BH2, 6, [(C5Me4H)2Sc][(μ-Ph)BPh3], 7, and [(C5Me5)2Sc][(μ-Ph)BPh3], 8. Structural information was also obtained on the borohydride derivatives, (C5Me4H)2Sc(μ-H)2BC8H14, 9, and (C5Me5)2Sc(μ-H)2BC8H14, 10, obtained from 9-borabicyclo(3.3.1)nonane (9-BBN) and (C5Me4R)2Sc(η3-C3H5), where R = H, 11; Me, 12. The preference of the metals for borohydride over tetraphenylborate binding was shown by the facile displacement of (BPh4)1− in 3, 4, 7, and 8 by (BH4)1− to make the respective borohydride complexes 1, 2, 5, and 6. These results are consistent with the fact that the borohydrides are not as useful as precursors in A2LnA′/M reductions of N2. An unusual structural isomer of [(C5Me4H)2Sc]2(μ-η2:η2-N2), 13′, was isolated from this study that shows the variations in ligand orientation that can occur in the solid state.

 Please wait while we load your content...
Please wait while we load your content...