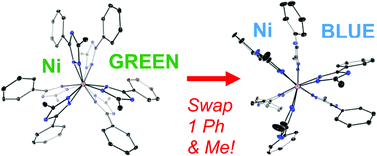
The tris(3-phenyl-5-methyl-1,2,4-triazolyl)borate (TtzPh,Me) ligand provides intermediate steric bulk and forms predominantly bis(ligand) complexes of the form M(TtzPh,Me)2 with first row divalent transition metals (1M, M = Zn, Cu, Ni, Co, Fe, Mn). Due to ligand field effects that are greatest with Ni and Cu, ligand rearrangement is favored with these metals and Cu(TtzPh,Me*)2 (1Cu*) and (TtzPh,Me*)Ni(TtzPh,Me) (1Ni*) were isolated by selective recrystallization and fully characterized (* indicates a rearranged Ttz ligand with Ph and Me in swapped positions in one triazole ring). For comparison with Co(TtzPh,Me)2, the less bulky analogs (TtzH,H)2Co (4) and (TtzMe,Me)2Co (5) were studied by NMR and EPR spectroscopy, and 5 was crystallographically characterized. These complexes allow for a study of how slight changes in structure and electron donor properties (for Ni and Cu), as well as dramatic changes in steric bulk (for Co), influence the physical properties; specifically there are significant changes in the UV-Vis, EPR and NMR spectra. Bis(ligand) complexes predominate with all metals, but (TtzPh,Me)Ni(OH2)Cl (2) and (TtzPh,Me)ZnBr (3) were also isolated and these show that TtzPh,Me is coordinatively flexible.

 Please wait while we load your content...
Please wait while we load your content...