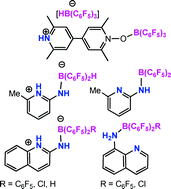
The lutidine derivative (2,6-Me2)(4-Bpin)C5H2N when combined with B(C6F5)3 yields a frustrated Lewis pair (FLP) which reacts with H2 to give the salt [(2,6-Me2)(4-Bpin)C5H2NH][HB(C6F5)3] (1). Similarly 2,2′-(C5H2(4,6-Me2)N)2 and (4,4′-(C5H2(4,6-Me2)N)2 were also combined with B(C6F5)3 and exposed to H2 to give [(2,2′-HN(2,6-Me2)C5H2C5H2(4,6-Me2)N][HB(C6F5)3] (2) and [(4,4′-HN(2,6-Me2)C5H2C5H2(2,6-Me2)N] [HB(C6F5)3] (3), respectively. The mono-pyridine-N-oxide 4,4′-N(2,6-Me2)C5H2C5H2(2,6-Me2)NO formed the adduct (4,4′-N(2,6-Me2)C5H2C5H2(2,6-Me2)NO)(B(C6F5)3) (4) which reacts further with B(C6F5)3 and H2 to give [(4,4′-HN(2,6-Me2)C5H2C5H2(2,6-Me2)NO)B(C6F5)3] [HB(C6F5)3] (5). In a related sense, 2-amino-6-CF3-C5H3N reacts with B(C6F5)3 to give (C5H3(6-CF3)NH)(2-NH(B(C6F5)3)) (6). Similarly, the species, 2-amino-quinoline, 8-amino-quinoline and 2-hydroxy-6-methyl-pyridine were reacted with B(C6F5)3 to give the products as (C9H6NH)(2-NHB(C6F5)3) (7), (C9H6N)(8-NH2B(C6F5)3) (8) and (C5H3(6-Me)NH)(2-OB(C6F5)3) (9), respectively; while 2-amino-6-picoline, 2-amino-6-CF3-pyridine, 2-amino-quinoline, 8-amino-quinoline and 2-hydroxy-6-methyl-pyridine react with ClB(C6F5)2 to give the species (C5H3(6-R)NH)(2-NH(ClB(C6F5)2)) (R = Me (10), R = CF3 (11)) (C9H6NH)(2-NH(ClB(C6F5)2)) (12), (C9H6N)(8-NH2ClB(C6F5)2) (13) and (C5H3(6-Me)NH)(2-OClB(C6F5)2) (14), respectively. In a similar manner, 2-amino-6-picoline and 2-amino-quinoline react with B(C6F5)2H to give (C5H3(6-Me)NH)(2-NH(HB(C6F5)2)) (15) and (C9H6NH)(2-NH(HB(C6F5)2)) (16). The corresponding reaction of 8-amino-quinoline yields (C9H6N)(8-NHB(C6F5)2) (17). In a similar fashion, reaction of 2-amino-6-CF3-pyridine resulted in the formation of (18) formulated as (C5H3(6-CF3)N)(2-NH(B(C6F5)2). Finally, treatment of 15 with iPrMgCl gave (C9H6N)(2-NH(B(C6F5)2)) (19). Crystallographic studies of 1, 2, 4, 6, 7, 10, 11, 12 and 15 are reported.

 Please wait while we load your content...
Please wait while we load your content...