Discrimination of fluorescence light-up effects induced by pH and metal ion chelation on a spirocyclic derivative of rhodamine B†
Abstract
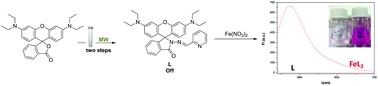
In the present work we describe the structure and the spectroscopic characterization of a spirocyclic derivative of a
- This article is part of the themed collection: Chemistry and applications of metal complexes

 Please wait while we load your content...
Please wait while we load your content...