The reactions of para-halo diaryl diselenides with halogens. A structural investigation of the CT compound (p-FC6H4)2Se2I2, and the first reported “RSeI3” compound, (p-ClC6H4)SeI·I2, which contains a covalent Se–I bond†‡
Abstract
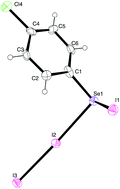
The reactions of the diaryl-diselenides (p-FC6H4)2Se2 and (p-ClC6H4)2Se2 with diiodine have been investigated. Species of stoichiometry “RSeI” are formed when the ratio employed is 1 : 1. The solid-state structure of “(p-FC6H4)SeI” has been determined, and shown to be a charge-transfer (CT) adduct, (p-FC6H4)2Se2I2, where the Se–Se bond is retained and the diiodine molecule interacts with only one of the selenium atoms. The Se–I bond in (p-FC6H4)2Se2I2 is 2.9835(12) Å, which is typical for a (10-I-2) Se–I–I CT system. When diiodine is reacted in a 3 : 1 ratio with (p-XC6H4)2Se2 (X = F, Cl) species of stoichiometry “RSeI3” are formed. The structure of “(p-ClC6H4)SeI3” reveals that this is not a selenium(IV) compound, but is better represented as a selenium(II) CT adduct, (p-ClC6H4)SeI·I2. The Se–I bond to the diiodine molecule is typical in magnitude for a CT adduct, Se–I: 2.8672(5) Å, whereas the other Se–I bond is much shorter, Se–I: 2.5590(6) Å, and is a genuine example of a rarely observed covalent Se–I bond, which appears to be stabilised by a weak Se⋯I interaction from a neighbouring iodine atom. The reaction of (p-ClC6H4)SeI with Ph3P results in the formation of a CT adduct, Ph3PSe(p-ClC6H4)I, which has a T-shaped geometry at selenium (10-Se-3). By contrast, the reaction of (p-FC6H4)SeI with Ph3P does not form an adduct, but results in the formation of Ph3PI2 and (p-FC6H4)2Se2.

 Please wait while we load your content...
Please wait while we load your content...