Protonreduction to hydrogen in biological and chemical systems
Abstract
In the drive to devise catalytic systems to convert solar energy into the energy of chemical bonds, chemists and electrochemists are seeking inspiration from our understanding of enzymes involved in bioenergetics. This is particularly true for generating molecular hydrogen from high energy electrons derived from solar driven
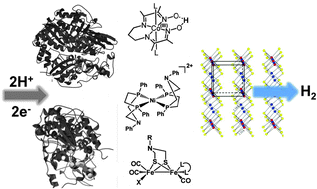
- This article is part of the themed collection: Electron Transfer Theory

 Please wait while we load your content...
Please wait while we load your content...