Chemical bonding in supermolecular flowers†
Abstract
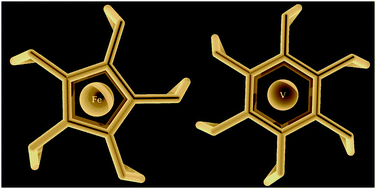
We report here a systematic study on the ability of molecular cages to bind (transition) metals. Starting from the superferrocenophane we investigate the incorporation of first-row transition metal (Sc–Zn) and alkaline-earth metal (Mg, Ca) double cations into these supermetallocenophane (super[5]phane) cages, and compare them with the corresponding metallocenes (Inorg. Chim. Acta, 2007, 360, 179). Moreover, we also investigate the binding of neutral and double-cationic metals inside supermetallocyclophane (super[6]phane) cages. The heterolytic and homolytic associations show preferences for different metals, and new metal-containing cages are proposed that should be viable candidates for synthesis.
- This article is part of the themed collection: Predicting new molecules by quantum chemical methods

 Please wait while we load your content...
Please wait while we load your content...