Oxidative dehydrogenation of cyclohexene on size selected subnanometer cobalt clusters: improved catalytic performance via evolution of cluster-assembled nanostructures
Abstract
The catalytic activity of
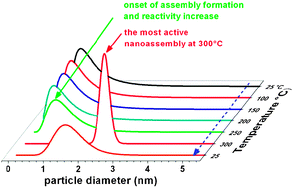
- This article is part of the themed collection: Structure and reactivity of small particles: from clusters to aerosols

 Please wait while we load your content...
Please wait while we load your content...