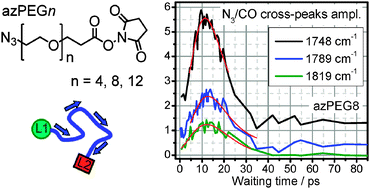
Dual-frequency relaxation-assisted two-dimensional infrared (RA 2DIR) spectroscopy was used to investigate energy transport in polyethylene glycol (PEG) oligomers of different length, having 0, 4, 8, and 12 repeating units and end-labeled with azido and succinimide ester moieties (azPEGn). The energy transport initiated by excitation of the N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N stretching mode of the azido group in azPEGn in CCl4 at ca. 2100 cm−1 was recorded by probing the C
N stretching mode of the azido group in azPEGn in CCl4 at ca. 2100 cm−1 was recorded by probing the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching modes (reporters) of the succinimide ester moiety. Sensitive to the excess energy delivered to the reporter modes, RA 2DIR permits observation of both the through-bond and through-solvent energy transport contributions. The cross-peak data involving the reporter modes with different thermal sensitivity and the data for mixtures of compounds permitted concluding that through-bond energy transport is the dominant mechanism for most cross peaks in all four azPEGn compounds. The through-bond energy transport time, evaluated as the waiting time at which the cross peak maximum is reached, was found to be linearly dependent on the chain length of up to 60 Å, suggesting a ballistic energy transport regime. The through-bond energy transport speed determined from the chain-length dependence of Tmax in CCl4 is found to be ca. 450 m s−1. The cross-peak amplitude at the maximum decays exponentially with the chain length; a characteristic decay distance is found to be 15.7 ± 1 Å. The cross-peak amplitude at zero waiting time, determined by the end-to-end distance distribution, is found to decay with the chain length (L) as ∼L−1.4, which is close to predictions of the free flight chain model. The match indicates that the end-group interaction does not strongly perturb the end-to-end distribution, which is close to the ideal random coil distribution with the Gaussian probability density.
O stretching modes (reporters) of the succinimide ester moiety. Sensitive to the excess energy delivered to the reporter modes, RA 2DIR permits observation of both the through-bond and through-solvent energy transport contributions. The cross-peak data involving the reporter modes with different thermal sensitivity and the data for mixtures of compounds permitted concluding that through-bond energy transport is the dominant mechanism for most cross peaks in all four azPEGn compounds. The through-bond energy transport time, evaluated as the waiting time at which the cross peak maximum is reached, was found to be linearly dependent on the chain length of up to 60 Å, suggesting a ballistic energy transport regime. The through-bond energy transport speed determined from the chain-length dependence of Tmax in CCl4 is found to be ca. 450 m s−1. The cross-peak amplitude at the maximum decays exponentially with the chain length; a characteristic decay distance is found to be 15.7 ± 1 Å. The cross-peak amplitude at zero waiting time, determined by the end-to-end distance distribution, is found to decay with the chain length (L) as ∼L−1.4, which is close to predictions of the free flight chain model. The match indicates that the end-group interaction does not strongly perturb the end-to-end distribution, which is close to the ideal random coil distribution with the Gaussian probability density.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N stretching mode of the
N stretching mode of the ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching modes (reporters) of the succinimide ester moiety. Sensitive to the excess energy delivered to the reporter modes, RA
O stretching modes (reporters) of the succinimide ester moiety. Sensitive to the excess energy delivered to the reporter modes, RA 

 Please wait while we load your content...
Please wait while we load your content...