The occurrence and representation of three-centre two-electron bonds in covalent inorganic compounds
Abstract
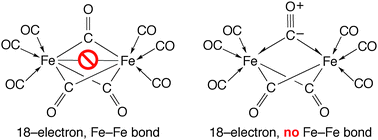
Although compounds that feature 3-centre 2-electron (3c–2e) bonds are well known, there has been no previous effort to classify the interactions according to the number of electrons that each atom contributes to the bond, in a manner analogous to the classification of 2-centre 2-electron (2c–2e) bonds as either normal covalent or dative covalent. This article provides an extension to the Covalent Bond Classification (CBC) method by categorizing 3c–2e interactions according to whether (i) the two electrons are provided by one or by two atoms and (ii) the central bridging atom provides two, one, or zero electrons. Class I 3c–2e bonds are defined as those in which two atoms each contribute one electron to the 3-centre orbital, while Class II 3c–2e bonds are defined as systems in which the pair of electrons are provided by a single atom. Class I and Class II 3c–2e interactions can be denoted by structure–bonding representations that employ the “half-arrow” notation, which also provides a convenient means to determine the electron count at a metal centre. In contrast to other methods of electron counting, this approach provides a means to predict metal–metal bond orders that are in accord with theory. For example, compounds that feature symmetrically bridging carbonyl ligands do not necessarily have to be described as “ketone derivatives” because carbon monoxide can also serve as an electron pair donor to two metal centres. This bonding description also provides a simple means to rationalize the theoretical predictions of the absence of M–M bonds in molecules such as Fe2(CO)9 and [CpFe(CO)2]2, which are widely misrepresented in textbooks as possessing M–M bonds.
- This article is part of the themed collection: Malcolm Green FRS: In celebration of his 80th Birthday

 Please wait while we load your content...
Please wait while we load your content...