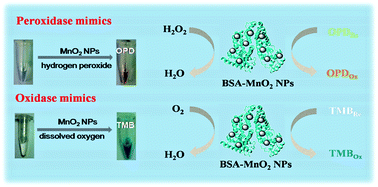
Inorganic nanomaterials that mimic enzymes are fascinating as they potentially have improved properties relative to native enzymes, such as greater resistance to extremes of pH and temperature and lower sensitivity to proteases. Although many artificial enzymes have been investigated, searching for highly-efficient and stable catalysts is still of great interest. In this paper, we first demonstrated that bovine serum albumin (BSA)-stabilized MnO2 nanoparticles (NPs) exhibited highly peroxidase-, oxidase-, and catalase-like activities. The activities of the BSA-MnO2 NPs were evaluated using the typical horseradish peroxidase (HRP) substrates o-phenylenediamine (OPD) and 3,3′,5,5′-tetramethylbenzidine (TMB) in the presence of either hydrogen peroxide or dissolved oxygen. These small-sized BSA-MnO2 NPs with good dispersion, solubility and biocompatibility exhibited typical Michaelis–Menten kinetics and high affinity for H2O2, OPD and TMB, indicating that BSA-MnO2 NPs can be used as satisfactory enzyme mimics. Based on these findings, BSA-MnO2 NPs were used as colorimetric immunoassay tags for the detection of goat anti-human IgG in place of HRP. The colorimetric immunoassay using BSA-MnO2 NPs has the advantages of being fast, robust, inexpensive, easily prepared and with no HRP and H2O2 being needed. These water-soluble BSA-MnO2 NPs may have promising potential applications in biotechnology, bioassays, and biomedicine.

 Please wait while we load your content...
Please wait while we load your content...