Comparative hydrolysis and plasma protein binding of cis-platin and carboplatin in human plasma in vitro†
Abstract
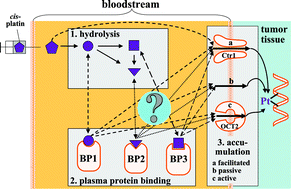
Platinum-based anti-cancer
* Corresponding authors
a
Department of Chemistry, University of Calgary, 2500 University Drive NW, Calgary, AB, Canada
E-mail:
jgailer@ucalgary.ca
Fax: +1 403-289-9488
Tel: +1 403-210-8899
b Alberta Children's Hospital, Pediatric Oncology, 2888 Shaganappi Trail N.W., AB, Canada
Platinum-based anti-cancer

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
