Real-time PCR of single bacterial cells on an array of adhering droplets
Abstract
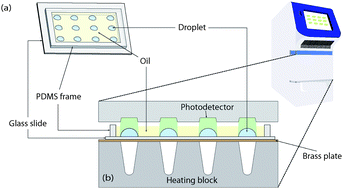
Real-time PCR at the single bacterial cell level is an indispensable tool to quantitatively reveal the heterogeneity of isogenetic cells. Conventional PCR platforms that utilize microtiter plates or PCR tubes have been widely used, but their large reaction volumes are not suited for sensitive single-cell analysis. Microfluidic devices provide high density, low volume PCR chambers, but they are usually expensive and require dedicated equipment to manipulate liquid and perform detection. To address these limitations, we developed an inexpensive chip-level device that is compatible with a commercial real-time PCR thermal cycler to perform quantitative PCR for single bacterial cells. The chip contains twelve surface-adhering droplets, defined by hydrophilic

 Please wait while we load your content...
Please wait while we load your content...