VEGF internalization is not required for VEGFR-2 phosphorylation in bioengineered surfaces with covalently linked VEGF†
Abstract
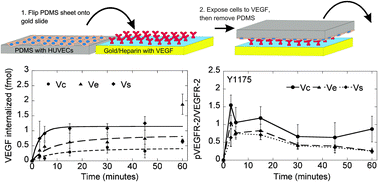
Vascular endothelial growth factor (VEGF) is known to activate proliferation, migration, and survival pathways in endothelial cells through phosphorylation of VEGF receptor-2 (VEGFR-2). VEGF has been incorporated into biomaterials through encapsulation, electrostatic sequestration, and covalent attachment, but the effect of these immobilization strategies on VEGF signaling has not been thoroughly investigated. Further, although growth factor internalization along with the receptor generally occurs in a physiological setting, whether this internalization is needed for receptor phosphorylation is not entirely clear. Here we show that VEGF covalently bound through a modified

 Please wait while we load your content...
Please wait while we load your content...