Microtopographical assembly of cardiomyocytes†
Abstract
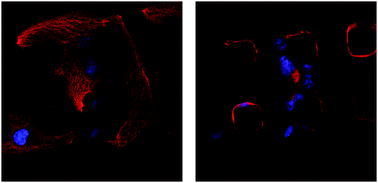
One of the central challenges in cardiac tissue engineering is the control of the assembly and organization of functional cardiac tissue. Maintenance of a three-dimensional tissue architecture is key to myocardial function in vivo, and a variety of studies hint that provision of topological cues within scaffolds can facilitate the engineering of functional myocardial tissue by promoting this architecture. To explore this possibility in an isolated and well-defined fashion, we have designed scaffolds of polydimethylsiloxane (PDMS) with microtopographic pillars (“micropegs”) to provide cells with defined structures with which to interact in three dimensions. We show that these surfaces permit HL-1 cardiomyocytes to grow, form myofibrillar structures and cell–cell adhesions, and beat spontaneously. Additionally, the cells and their nuclei interact with the full length of the micropegs, indicating that the micropegs promote a three-dimensional cytoarchitecture in the context of a two-dimensional scaffold. We also show that the number of cells interacting with a micropeg can be controlled by manipulating incubation time, micropeg spatial arrangement, or micropeg diameter. Western blots reveal that the expression of the junctional markers N-cadherin and connexin 43 is upregulated in the presence of specific arrangements of micropegs, suggesting that micropegs can enhance cardiomyocyte function. Together, these data show that microtopography can be used to provide three-dimensional adhesion and control the assembly of functional cardiac tissue on a two-dimensional surface.
 Please wait while we load your content...
Please wait while we load your content...