Abstract
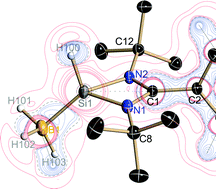
A stable silicon(II) monohydride is accomplished through a covalent shared interaction of the silylene lone-pair and a sp3-hybridized boron atom of the Lewis acidic BH3. Experimental charge density investigations reveal a central positively charged silicon atom bound to a negatively charged hydrogen atom. The positively charged H–Si–BH3 moiety is coordinated by the lone-pairs of electrons of the benzamidinate

 Please wait while we load your content...
Please wait while we load your content...