Structural resolution of carbohydrate positional and structural isomers based on gas-phase ion mobility-mass spectrometry†‡
Abstract
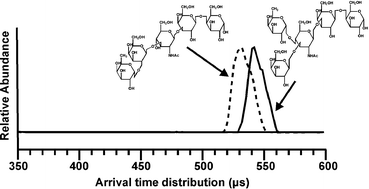
This report describes the rapid characterization of positional and structural carbohydrate isomers based on structural separations provided by ion mobility-

 Please wait while we load your content...
Please wait while we load your content...