Molecular organization of hydrophobic molecules and co-adsorbed water in SBA-15 ordered mesoporous silica material†
Abstract
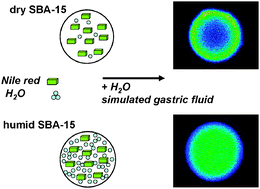
The purpose of this study was to improve our understanding of the molecular organization of hydrophobic guest molecules in the presence of co-adsorbed

 Please wait while we load your content...
Please wait while we load your content...