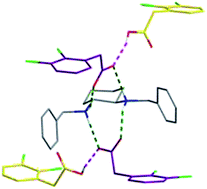
trans-N,N′-Dibenzyldiaminocyclohexane (B) crystallizes with 2,3-dichlorophenylacetic acid (AH); the crystal structure, resolved by using crystal X-ray diffraction, revealed an odd stoichiometry composed of H2B2+, two A− and two AH forming an unexpected hybrid salt–cocrystal. As this compound is a stable conglomerate (i.e. every single crystal contains the enantiomerically pure cation H2B2+, RR or SS), several preferential crystallization attempts (AS3PC) were performed in methanol and in THF and gave unexpected final enantiomeric excesses greater than 20% for the entrainment in methanol. These results suggest that the crystal growth mechanism preferentially involves building units composed of [H2B2+; 2A− and 2AH] or reconstruction of some crystal interfaces rather than a layer by layer construction.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...