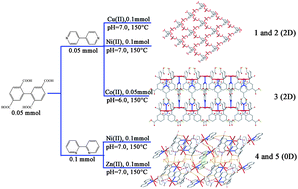
Five new metal complexes based on H4bptc: {[Cu2(bptc)(H2O)6]·11H2O}n (1), {[Ni2(bptc)(H2O)6]·11H2O}n (2), {[Co2(H2bptc)2(4,4′-bpy)2(H2O)2]·4H2O}n (3), [Ni2(bptc)(2,2′-bpy)2(H2O)6]·2H2O (4), [Zn2(bptc)(2,2′-bpy)2(H2O)6]·3H2O (5) (bptc = biphenyl-2,5,2′,5′-tetracarboxylic acid, 2,2′-bpy = 2,2′-bipyridine, 4,4′-bpy = 4,4′-bipyridine) have been synthesized and structurally characterized by elemental analysis, IR spectra, powder X-ray diffraction, thermogravimetric analysis and X-ray crystallography. Complex 1 features a 2D sheet structure with (4, 4) grid units. The Cu(II) center in 1 is penta-coordinated in a square-pyramidal geometry. Complex 2 is isostructural with 1 excluding the metal ions. Complex 3 shows a 2D layer structure with rectangular windows. There exist parallel left- and right-handed helical chains in adjacent 2D sheets in complexes 1–3. Complex 4 exhibits a 3D supramolecular structure with a butterfly-shaped unit cell. Complexes 5 and 4 are isomers excluding the lattice water molecules. The non-covalent interactions (O–H⋯O, C–H⋯O, C–H⋯π and π⋯π interactions) extend complexes 1–5 into 3D supramolecular networks. In addition, the photoluminescent properties for 1–5 and magnetic properties for 1, 3 have been investigated, respectively. Complexes 1 and 3 exhibit weak antiferromagnetic behaviour.

 Please wait while we load your content...
Please wait while we load your content...