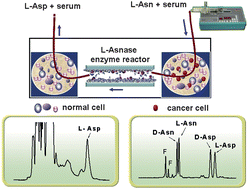
The study of enzyme immobilization using an extracorporeal shunt system is essential to eliminate the side effects of L-asparaginase (L-Asnase; including hepatic toxicity, allergic reaction, pancreatitis, central nervous system toxicity and decreased synthesis of blood clotting factors) when it was applied as an anticancer drug given directly to patients by injection. Thus, the novel monolith and coating enzymatic reactors of L-asparaginase were provided in this assay and a microchip electrophoresis–laser induced fluorescence (MCE–LIF) method was set up for the enzyme kinetics study. The enzymatic reactors would be a promising in vitro therapeutic method in an extracorporeal shunt system for acute lymphoblastic leukemia (ALL) treatment. For the first time, L-asparaginase was covalently bound to the polymer monolith and coating in the capillary and the activity characteristics of these enzymatic microreactors have been probed by Michaelis–Menten kinetic constants. Meanwhile, the D,L-amino acids were chirally separated using microchip electrophoresis with a laser induced detector and D,L-aspartic acid (D,L-Asp) were tested for the L-asparaginase enzymatic reactor kinetics study. Furthermore, human serum adding with L-asparagine (L-Asn) as the sample was hydrolyzed by the enzymatic microreactors. The results demonstrated that the developed enzymatic microreactor of L-asparaginase would be a potential therapeutic protocol for ALL treatment.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...