Selectivity enhancement in functionalization of C–H bonds: A review
Abstract
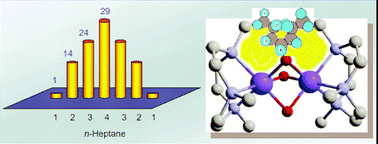
The selectivity is an extremely important characteristic of a chemical reaction. This review deals mainly with supramolecular and nano-chemical approaches to the problem of selectivity enhancement in various functionalizations of C–H compounds.
 Please wait while we load your content...
Please wait while we load your content...