On the importance of simultaneous infrared/fiber-optic temperature monitoring in the microwave-assisted synthesis of ionic liquids†‡
Abstract
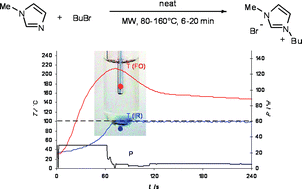
The temperature profiles obtained from both an external infrared and internal fiber-optic sensor were compared for heating and synthesizing the ionic liquid

 Please wait while we load your content...
Please wait while we load your content...